Understanding the Boron Electron Structure is fundamental to grasping the properties and behaviors of this essential element. Boron, with the atomic number 5, is a metalloid that plays a crucial role in various industrial and technological applications. Its unique electron configuration contributes to its distinctive chemical and physical properties, making it a subject of interest in both academic and practical settings.
Introduction to Boron
Boron is the fifth element in the periodic table, positioned between beryllium and carbon. It is classified as a metalloid, exhibiting properties of both metals and non-metals. Boron's atomic number is 5, which means it has 5 protons and 5 electrons. The element is known for its hardness and high melting point, making it valuable in materials science and engineering.
Electron Configuration of Boron
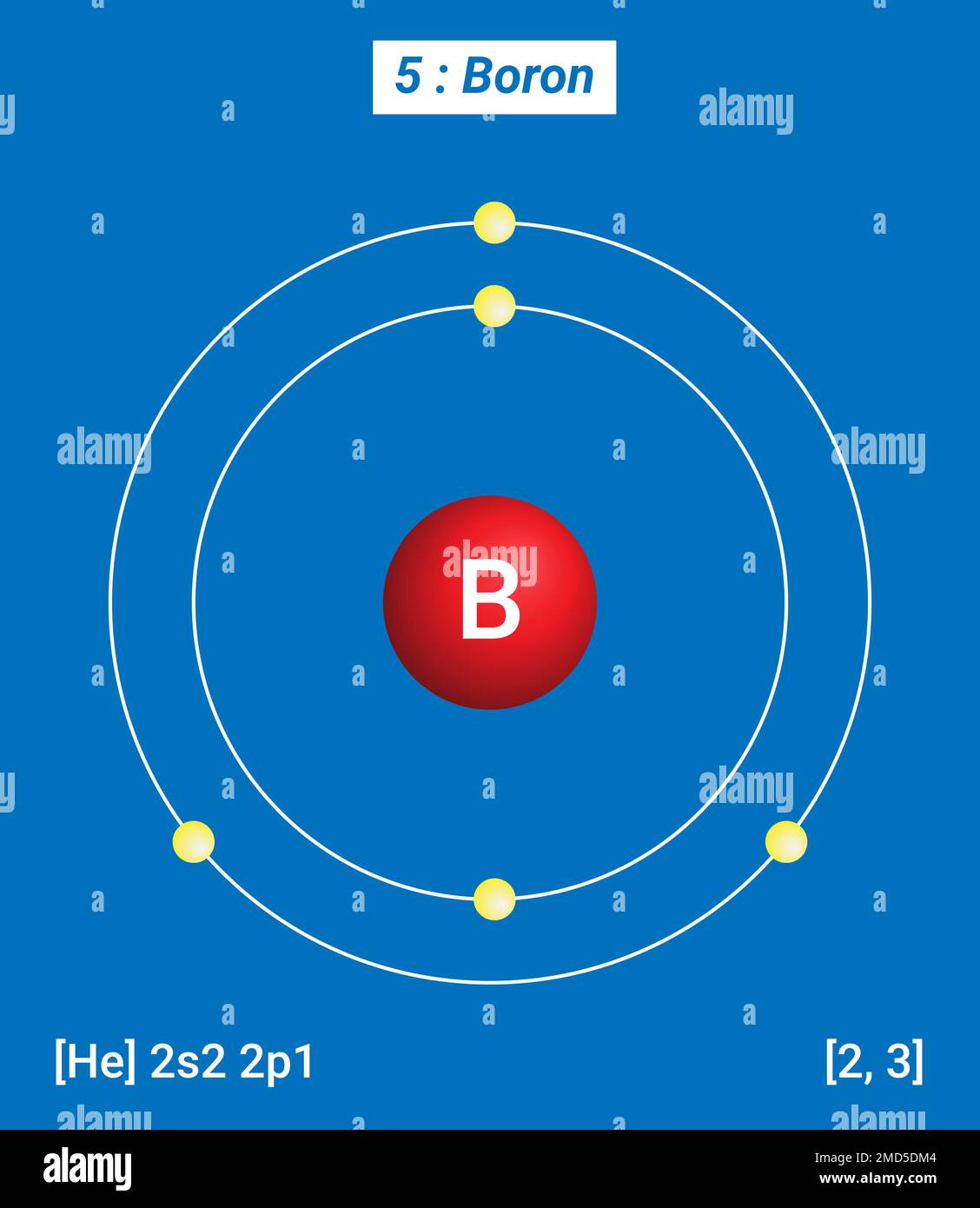
The Boron Electron Structure can be understood by examining its electron configuration. The electron configuration of an atom describes the distribution of electrons in its atomic orbitals. For boron, the electron configuration is:
1s2 2s2 2p1
This configuration indicates that boron has:
- 2 electrons in the 1s orbital
- 2 electrons in the 2s orbital
- 1 electron in the 2p orbital
This distribution of electrons in the orbitals is crucial for understanding boron's chemical behavior and reactivity.
Boron's Position in the Periodic Table
Boron is located in Group 13 (formerly known as Group IIIA) of the periodic table. This group includes elements like aluminum, gallium, indium, and thallium. Elements in this group have three electrons in their outermost shell, which gives them a valence of +3. However, boron's unique Boron Electron Structure allows it to form compounds with a variety of oxidation states, including +1 and +2.
Chemical Properties of Boron
The chemical properties of boron are largely determined by its Boron Electron Structure. Some key chemical properties include:
- Electronegativity: Boron has an electronegativity of 2.04 on the Pauling scale, making it moderately electronegative.
- Ionization Energy: The first ionization energy of boron is 800.6 kJ/mol, which is relatively high compared to other elements in its group.
- Atomic Radius: Boron has a small atomic radius of 82 pm, contributing to its high density and hardness.
These properties make boron a versatile element in various chemical reactions and industrial applications.
Boron Compounds
Boron forms a wide range of compounds due to its unique Boron Electron Structure. Some of the most notable boron compounds include:
| Compound | Formula | Uses |
|---|---|---|
| Boron Trifluoride | BF3 | Used as a catalyst in organic synthesis and in the production of polymers. |
| Borax | Na2B4O7·10H2O | Used in laundry detergents, glassmaking, and as a flux in metallurgy. |
| Boron Nitride | BN | Used as a high-temperature lubricant and in the production of advanced ceramics. |
| Boron Carbide | B4C | Used in armor plating, cutting tools, and as a neutron absorber in nuclear reactors. |
These compounds highlight the versatility of boron in various industrial and technological applications.
Applications of Boron
The unique properties of boron, stemming from its Boron Electron Structure, make it valuable in numerous applications. Some of the key areas where boron is used include:
- Materials Science: Boron is used in the production of high-strength materials, such as boron carbide and boron nitride, which are used in cutting tools, armor plating, and advanced ceramics.
- Agriculture: Boron is an essential micronutrient for plants, playing a crucial role in cell wall formation and membrane function. It is often added to fertilizers to ensure adequate plant growth.
- Medicine: Boron compounds are used in various medical applications, including as antiseptics and in the treatment of certain types of cancer.
- Energy: Boron is used in nuclear reactors as a neutron absorber to control the fission process. It is also used in the production of boron-doped silicon for solar cells.
These applications demonstrate the wide-ranging utility of boron in modern technology and industry.
📝 Note: The properties and applications of boron are continually being explored, leading to new discoveries and innovations in various fields.
Boron in the Environment
Boron is found naturally in the Earth's crust, primarily in the form of borax and other boron minerals. It is also present in seawater and some plants. The environmental impact of boron is generally low, but excessive amounts can be toxic to plants and animals. Boron is essential for plant growth, but high concentrations can inhibit growth and cause toxicity symptoms.
Boron's environmental presence and its role in ecosystems are influenced by its Boron Electron Structure, which affects its chemical reactivity and mobility in the environment.
Future Prospects of Boron Research
The study of boron and its Boron Electron Structure continues to be an active area of research. Scientists are exploring new applications for boron in materials science, energy storage, and medicine. Advances in boron research have the potential to lead to breakthroughs in various fields, including:
- Nanotechnology: Boron-based nanomaterials are being developed for use in electronics, sensors, and energy storage devices.
- Catalysis: Boron compounds are being studied for their potential as catalysts in chemical reactions, offering more efficient and sustainable processes.
- Biomedicine: Boron neutron capture therapy (BNCT) is a promising cancer treatment that uses boron compounds to target and destroy cancer cells.
These areas of research highlight the ongoing importance of understanding boron's unique properties and applications.
Boron’s unique Boron Electron Structure makes it a fascinating element to study, with a wide range of applications in modern technology and industry. Its properties and behaviors are influenced by its electron configuration, which determines its chemical reactivity and physical characteristics. As research continues, new discoveries and innovations in boron science are likely to emerge, further expanding its utility and importance in various fields.
Related Terms:
- ground state electron configuration boron
- full electron configuration of boron
- boron electron shell diagram
- boron electron configuration orbital diagram
- electronic configuration for boron
- boron electron configuration diagram