Methane, a colorless and odorless gas, is a significant component of natural gas and a potent greenhouse gas. Understanding the flammable range methane is crucial for safety and environmental management. This post delves into the properties of methane, its flammable range, detection methods, safety measures, and environmental impact.
Properties of Methane
Methane (CH₄) is the simplest hydrocarbon, consisting of one carbon atom and four hydrogen atoms. It is highly flammable and can form explosive mixtures with air. Key properties include:
- Molecular Weight: 16.04 g/mol
- Boiling Point: -161.5°C (-258.7°F)
- Melting Point: -182.5°C (-296.5°F)
- Density: 0.716 kg/m³ at 0°C
- Flammability: Highly flammable in air
Understanding the Flammable Range of Methane
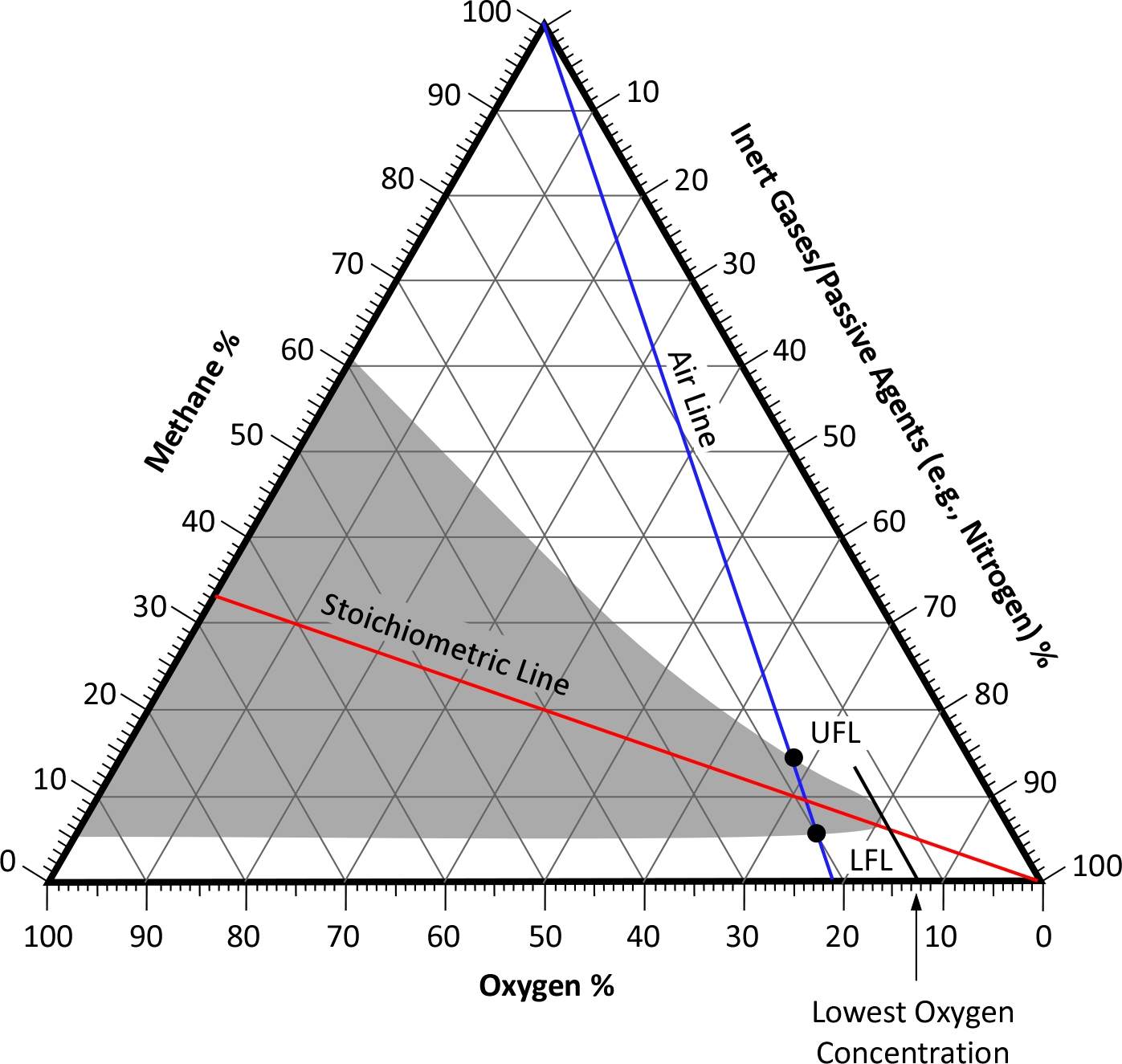
The flammable range methane refers to the concentration range within which methane can ignite and burn in the presence of an ignition source. This range is typically expressed as a percentage of methane in air. For methane, the flammable range is between 5% and 15% by volume. This means that a mixture of methane and air can ignite and burn if the methane concentration falls within this range.
Factors Affecting the Flammable Range
Several factors can influence the flammable range of methane:
- Temperature: Higher temperatures can widen the flammable range.
- Pressure: Increased pressure can also affect the flammable range.
- Oxygen Concentration: The presence of other gases, such as oxygen, can alter the flammable range.
- Presence of Inert Gases: Inert gases like nitrogen or carbon dioxide can narrow the flammable range.
Detection Methods for Methane
Detecting methane is essential for safety and environmental monitoring. Various methods are used to detect methane, including:
- Gas Detectors: Portable and fixed gas detectors use sensors to measure methane concentrations in the air.
- Infrared Spectroscopy: This method uses infrared light to detect methane molecules.
- Catalytic Bead Sensors: These sensors use a catalytic reaction to detect methane.
- Semiconductor Sensors: These sensors change their electrical resistance in the presence of methane.
Safety Measures for Handling Methane
Handling methane requires strict safety measures to prevent accidents and environmental damage. Key safety measures include:
- Ventilation: Ensure proper ventilation to prevent the accumulation of methane.
- Gas Detection Systems: Install gas detection systems to monitor methane levels continuously.
- Emergency Shutdown Systems: Implement emergency shutdown systems to stop methane flow in case of a leak.
- Personal Protective Equipment (PPE): Use appropriate PPE, including respirators and protective clothing.
- Training: Provide regular training for personnel on methane handling and emergency procedures.
Environmental Impact of Methane
Methane is a potent greenhouse gas with a global warming potential 25 times greater than carbon dioxide over a 100-year period. Its environmental impact includes:
- Climate Change: Methane contributes significantly to global warming.
- Air Pollution: Methane can react with other pollutants to form ground-level ozone, a harmful air pollutant.
- Ozone Depletion: Methane can indirectly contribute to the depletion of the ozone layer.
Regulations and Standards for Methane Management
Various regulations and standards govern the management of methane to ensure safety and environmental protection. Key regulations include:
- OSHA Regulations: The Occupational Safety and Health Administration (OSHA) sets standards for workplace safety, including methane handling.
- EPA Regulations: The Environmental Protection Agency (EPA) regulates methane emissions from various sources, including oil and gas operations.
- International Standards: Organizations like the International Organization for Standardization (ISO) provide guidelines for methane management.
Case Studies: Methane Leaks and Their Consequences
Methane leaks can have severe consequences, as illustrated by the following case studies:
| Case Study | Location | Cause | Consequences |
|---|---|---|---|
| Aliso Canyon Gas Leak | Porter Ranch, California | Faulty well casing | Evacuation of thousands of residents, significant environmental impact |
| San Bruno Pipeline Explosion | San Bruno, California | Corroded pipeline | 8 deaths, 58 injuries, extensive property damage |
| Deepwater Horizon Oil Spill | Gulf of Mexico | Blowout preventer failure | 11 deaths, massive oil and methane release, environmental disaster |
📌 Note: These case studies highlight the importance of proper methane management and the potential consequences of neglecting safety measures.
Future Trends in Methane Management
Advancements in technology and increased awareness are driving future trends in methane management. Key trends include:
- Advanced Detection Technologies: Development of more sensitive and accurate methane detection technologies.
- Remote Monitoring: Use of satellite and drone technology for remote monitoring of methane emissions.
- Regulatory Enhancements: Strengthening of regulations and standards for methane management.
- Public Awareness: Increased public awareness and education on the dangers of methane and the importance of proper management.
In conclusion, understanding the flammable range methane is vital for ensuring safety and minimizing environmental impact. By implementing proper detection methods, safety measures, and regulatory compliance, we can effectively manage methane and mitigate its risks. The environmental impact of methane underscores the need for continued research and innovation in methane management technologies. Through collective efforts, we can address the challenges posed by methane and work towards a safer and more sustainable future.
Related Terms:
- lower flammable limit for methane
- is methane dangerous
- flammable gases chart
- flammable range chart
- flammable gas range chart
- lower flammability limit chart