Understanding the fundamental differences between Molecular Vs Ionic Compound is crucial for anyone delving into the world of chemistry. These two types of compounds exhibit distinct properties and behaviors due to their unique bonding mechanisms. This blog post will explore the characteristics, formation, and applications of molecular and ionic compounds, providing a comprehensive overview to help you grasp these essential concepts.
Understanding Molecular Compounds
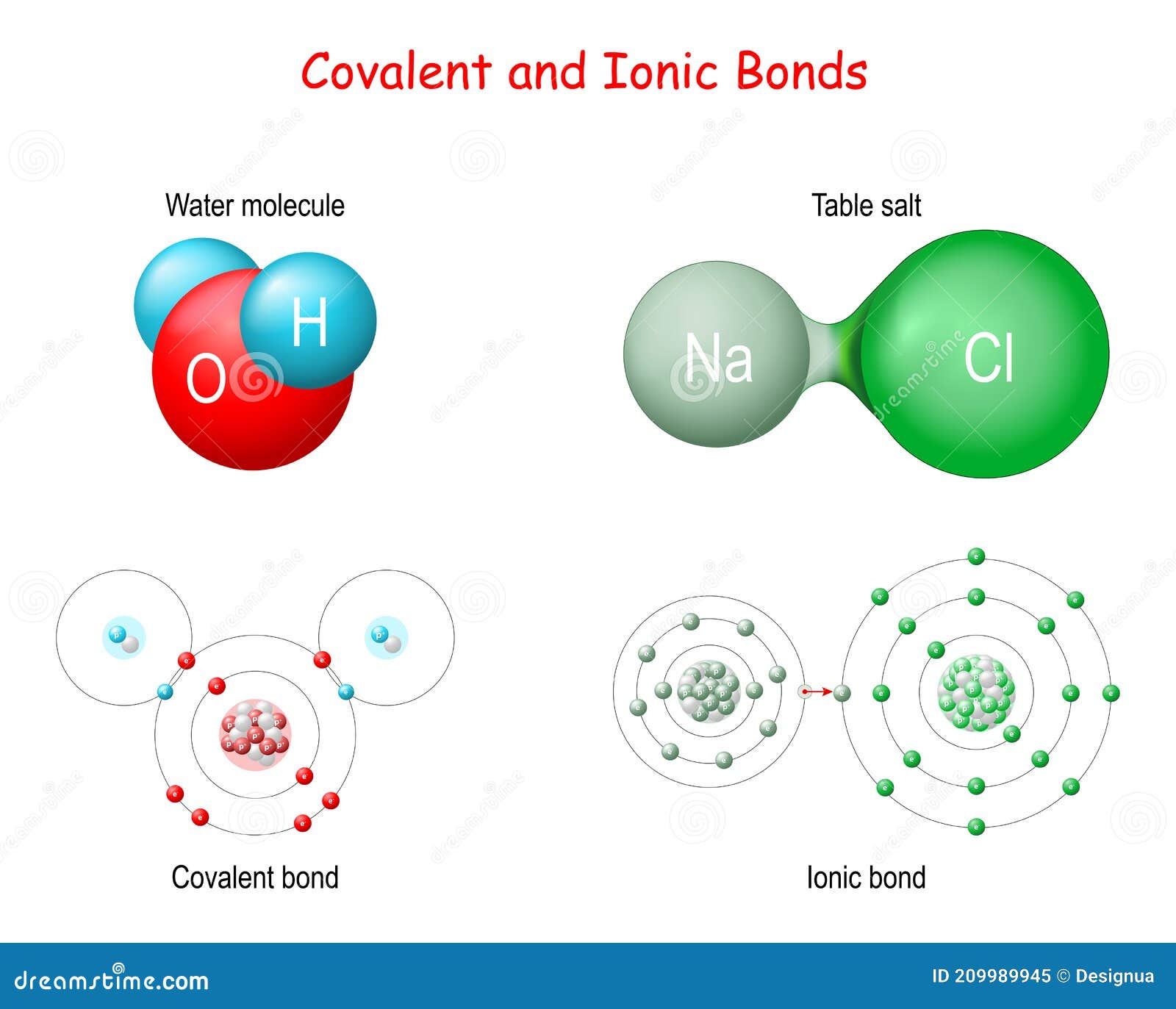
Molecular compounds are formed when atoms share electrons through covalent bonds. This sharing allows each atom to achieve a stable electron configuration, similar to that of a noble gas. The resulting molecules can be simple, consisting of just two atoms, or complex, with many atoms bonded together.
Characteristics of Molecular Compounds
Molecular compounds have several key characteristics:
- Low Melting and Boiling Points: Due to the relatively weak intermolecular forces, molecular compounds typically have low melting and boiling points.
- Poor Electrical Conductivity: Since there are no free ions or electrons, molecular compounds do not conduct electricity in their solid or liquid states.
- Solubility: Molecular compounds are often soluble in non-polar solvents but insoluble in polar solvents like water.
Examples of Molecular Compounds
Some common examples of molecular compounds include:
- Water (H2O)
- Carbon Dioxide (CO2)
- Methane (CH4)
- Glucose (C6H12O6)
Understanding Ionic Compounds
Ionic compounds are formed when atoms transfer electrons to achieve a stable electron configuration. This transfer results in the formation of positively charged ions (cations) and negatively charged ions (anions), which are held together by strong electrostatic forces known as ionic bonds.
Characteristics of Ionic Compounds
Ionic compounds have several distinctive characteristics:
- High Melting and Boiling Points: The strong electrostatic forces between ions result in high melting and boiling points.
- Good Electrical Conductivity: In their molten or dissolved states, ionic compounds conduct electricity due to the presence of free ions.
- Solubility: Ionic compounds are often soluble in polar solvents like water but insoluble in non-polar solvents.
Examples of Ionic Compounds
Some common examples of ionic compounds include:
- Sodium Chloride (NaCl)
- Potassium Bromide (KBr)
- Calcium Carbonate (CaCO3)
- Magnesium Sulfate (MgSO4)
Comparing Molecular Vs Ionic Compound
To better understand the differences between molecular and ionic compounds, let's compare their key properties in a table:
| Property | Molecular Compounds | Ionic Compounds |
|---|---|---|
| Bonding | Covalent (electron sharing) | Ionic (electron transfer) |
| Melting and Boiling Points | Low | High |
| Electrical Conductivity | Poor | Good (in molten or dissolved states) |
| Solubility | Soluble in non-polar solvents | Soluble in polar solvents |
| Examples | H2O, CO2, CH4 | NaCl, KBr, CaCO3 |
💡 Note: The table above provides a quick reference for the key differences between molecular and ionic compounds. Understanding these differences is essential for predicting the behavior of various substances in chemical reactions.
Applications of Molecular and Ionic Compounds
Both molecular and ionic compounds have numerous applications in various industries. Understanding their properties allows scientists and engineers to harness their unique characteristics for specific purposes.
Applications of Molecular Compounds
Molecular compounds are widely used in:
- Pharmaceuticals: Many drugs are molecular compounds designed to interact with specific biological targets.
- Food Industry: Molecular compounds like sugars and fats are essential components of food.
- Chemical Industry: Molecular compounds are used as solvents, fuels, and raw materials for various chemical processes.
Applications of Ionic Compounds
Ionic compounds are extensively used in:
- Construction: Ionic compounds like cement and concrete are crucial for building infrastructure.
- Medicine: Ionic compounds are used in medications, such as antacids and laxatives.
- Agriculture: Ionic compounds like fertilizers provide essential nutrients for plant growth.
Formation of Molecular and Ionic Compounds
Understanding how molecular and ionic compounds form is essential for grasping their properties and behaviors. The formation processes for these compounds differ significantly due to their distinct bonding mechanisms.
Formation of Molecular Compounds
Molecular compounds form through covalent bonding, where atoms share electrons to achieve stable electron configurations. The process involves:
- Electron Sharing: Atoms share electrons to fill their outer shells.
- Molecule Formation: Shared electrons form covalent bonds, creating molecules.
- Stable Configuration: The resulting molecules have stable electron configurations, similar to noble gases.
Formation of Ionic Compounds
Ionic compounds form through ionic bonding, where atoms transfer electrons to achieve stable electron configurations. The process involves:
- Electron Transfer: One atom donates electrons, becoming a cation, while another atom accepts electrons, becoming an anion.
- Ion Formation: The transfer of electrons results in the formation of positively and negatively charged ions.
- Electrostatic Attraction: Oppositely charged ions attract each other, forming an ionic bond.
💡 Note: The formation of molecular and ionic compounds involves different mechanisms, leading to their unique properties and behaviors. Understanding these mechanisms is crucial for predicting chemical reactions and designing new materials.
Conclusion
In summary, molecular and ionic compounds exhibit distinct properties due to their unique bonding mechanisms. Molecular compounds, formed through covalent bonding, have low melting and boiling points, poor electrical conductivity, and are soluble in non-polar solvents. In contrast, ionic compounds, formed through ionic bonding, have high melting and boiling points, good electrical conductivity in molten or dissolved states, and are soluble in polar solvents. Understanding these differences is essential for predicting the behavior of various substances in chemical reactions and harnessing their unique characteristics for specific applications.
Related Terms:
- different between ionic and molecular
- molecular vs ionic compound naming
- ionic vs molecular covalent compounds
- identify ionic vs molecular compounds
- molecular formula vs ionic
- compared with ionic compounds molecular