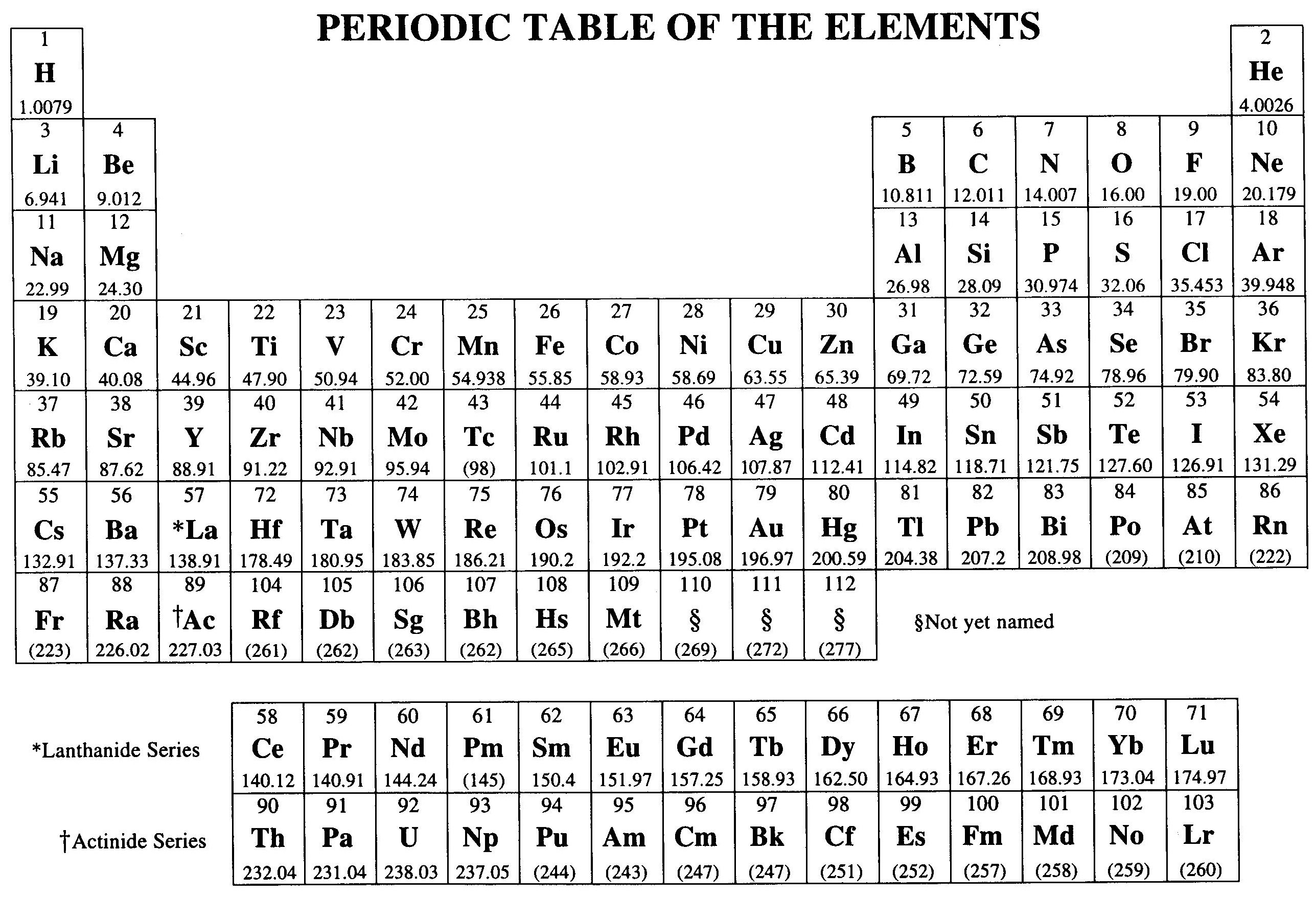
Chemistry is a fascinating field that delves into the properties and behaviors of matter at the atomic and molecular levels. One of the fundamental tools used in chemistry is the Reference Table Chemistry, which provides essential data and information that chemists rely on for their experiments and research. This table is a comprehensive resource that includes various chemical properties, constants, and formulas that are crucial for understanding and predicting chemical reactions.
Understanding the Reference Table Chemistry
The Reference Table Chemistry is a compilation of key data points that are essential for chemists and students alike. It includes information such as atomic weights, electronegativity values, ionization energies, and more. This table serves as a quick reference guide, allowing chemists to access important information without having to look it up in multiple sources.
Key Components of the Reference Table Chemistry
The Reference Table Chemistry is divided into several sections, each focusing on different aspects of chemical data. Some of the key components include:
- Atomic Weights: The atomic weight of an element is the average mass of its atoms, typically expressed in atomic mass units (amu). This information is crucial for calculating the molar mass of compounds.
- Electronegativity: Electronegativity is a measure of an atom’s ability to attract electrons towards itself in a chemical bond. This property is essential for predicting the polarity of bonds and the reactivity of compounds.
- Ionization Energies: Ionization energy is the amount of energy required to remove an electron from an atom or ion. This data is important for understanding the reactivity of elements and the formation of ions.
- Electron Affinities: Electron affinity is the energy released when an electron is added to a neutral atom or molecule to form a negative ion. This property is crucial for understanding the stability of anions and the reactivity of elements.
- Standard Reduction Potentials: Standard reduction potentials are measures of the tendency of a chemical species to gain electrons and be reduced. This information is essential for predicting the direction of redox reactions.
Importance of the Reference Table Chemistry
The Reference Table Chemistry plays a vital role in various aspects of chemical research and education. Here are some of the key reasons why it is so important:
- Educational Tool: For students, the Reference Table Chemistry is an invaluable resource for learning and understanding chemical concepts. It provides a quick and easy way to access essential data, making it easier to solve problems and understand complex chemical reactions.
- Research Aid: For researchers, the Reference Table Chemistry is a crucial tool for designing experiments and interpreting results. It allows chemists to quickly access the data they need to make informed decisions and predictions.
- Industrial Applications: In industrial settings, the Reference Table Chemistry is used to optimize processes and ensure the safety and efficiency of chemical reactions. It helps in selecting the right reagents, predicting reaction outcomes, and troubleshooting issues.
How to Use the Reference Table Chemistry
Using the Reference Table Chemistry effectively requires understanding how to navigate its various sections and interpret the data. Here are some steps to help you get the most out of this resource:
- Identify the Element or Compound: Start by identifying the element or compound you are interested in. This could be based on its name, symbol, or atomic number.
- Locate the Relevant Section: Once you have identified the element or compound, locate the relevant section in the Reference Table Chemistry. This could be the section on atomic weights, electronegativity, ionization energies, etc.
- Interpret the Data: Carefully read and interpret the data provided in the table. Make sure you understand what each value represents and how it relates to the chemical properties of the element or compound.
- Apply the Data: Use the data to solve problems, design experiments, or make predictions. For example, you might use the atomic weight to calculate the molar mass of a compound or the electronegativity to predict the polarity of a bond.
📝 Note: Always double-check the data in the Reference Table Chemistry to ensure accuracy, as slight variations can affect the outcomes of your experiments or calculations.
Common Applications of the Reference Table Chemistry
The Reference Table Chemistry has a wide range of applications in various fields of chemistry. Some of the most common applications include:
- Stoichiometry: The Reference Table Chemistry is essential for stoichiometry calculations, which involve determining the quantities of reactants and products in a chemical reaction. Atomic weights and molar masses are crucial for these calculations.
- Acid-Base Chemistry: Electronegativity and electron affinity values are important for understanding the behavior of acids and bases. These properties help predict the strength of acids and bases and their reactivity.
- Redox Reactions: Standard reduction potentials are used to predict the direction of redox reactions and to calculate cell potentials in electrochemical cells.
- Organic Chemistry: In organic chemistry, the Reference Table Chemistry is used to understand the reactivity of organic compounds, predict reaction mechanisms, and design synthetic routes.
Example of a Reference Table Chemistry
Below is an example of a simplified Reference Table Chemistry that includes some of the key data points for a few elements:
| Element | Symbol | Atomic Number | Atomic Weight (amu) | Electronegativity | Ionization Energy (kJ/mol) | Electron Affinity (kJ/mol) |
|---|---|---|---|---|---|---|
| Hydrogen | H | 1 | 1.008 | 2.20 | 1312 | 73 |
| Helium | He | 2 | 4.0026 | N/A | 2372 | N/A |
| Lithium | Li | 3 | 6.941 | 0.98 | 520 | 59.6 |
| Beryllium | Be | 4 | 9.0122 | 1.57 | 899 | N/A |
| Boron | B | 5 | 10.81 | 2.04 | 801 | 27 |
This table provides a quick reference for the atomic weights, electronegativity, ionization energies, and electron affinities of hydrogen, helium, lithium, beryllium, and boron. This information is essential for understanding the chemical properties and behaviors of these elements.
Advanced Uses of the Reference Table Chemistry
Beyond basic applications, the Reference Table Chemistry can be used for more advanced purposes. For example, it can help in the design of complex chemical reactions, the synthesis of new compounds, and the development of new materials. Researchers often use the data in the Reference Table Chemistry to predict the outcomes of experiments and to optimize reaction conditions.
In industrial settings, the Reference Table Chemistry is used to ensure the safety and efficiency of chemical processes. It helps in selecting the right reagents, predicting reaction outcomes, and troubleshooting issues. For example, in the pharmaceutical industry, the Reference Table Chemistry is used to design and synthesize new drugs, ensuring that they are safe and effective.
In environmental chemistry, the Reference Table Chemistry is used to understand the behavior of pollutants and to develop strategies for their remediation. It helps in predicting the reactivity of pollutants and their potential impact on the environment.
In materials science, the Reference Table Chemistry is used to design and develop new materials with specific properties. It helps in understanding the chemical and physical properties of materials and in predicting their behavior under different conditions.
In analytical chemistry, the Reference Table Chemistry is used to develop and optimize analytical methods. It helps in understanding the chemical properties of analytes and in selecting the right reagents and conditions for analysis.
In biochemistry, the Reference Table Chemistry is used to understand the chemical properties of biological molecules and to design and synthesize new biomolecules. It helps in predicting the behavior of biomolecules and in developing new therapeutic agents.
In physical chemistry, the Reference Table Chemistry is used to understand the fundamental principles of chemical reactions and to develop new theories and models. It helps in predicting the behavior of chemical systems and in designing new experiments.
In inorganic chemistry, the Reference Table Chemistry is used to understand the chemical properties of inorganic compounds and to design and synthesize new inorganic materials. It helps in predicting the behavior of inorganic compounds and in developing new applications.
In organic chemistry, the Reference Table Chemistry is used to understand the chemical properties of organic compounds and to design and synthesize new organic molecules. It helps in predicting the behavior of organic compounds and in developing new synthetic routes.
In polymer chemistry, the Reference Table Chemistry is used to understand the chemical properties of polymers and to design and synthesize new polymeric materials. It helps in predicting the behavior of polymers and in developing new applications.
In nuclear chemistry, the Reference Table Chemistry is used to understand the chemical properties of radioactive elements and to design and synthesize new radioactive materials. It helps in predicting the behavior of radioactive elements and in developing new applications.
In electrochemistry, the Reference Table Chemistry is used to understand the chemical properties of electrochemical systems and to design and optimize electrochemical cells. It helps in predicting the behavior of electrochemical systems and in developing new applications.
In photochemistry, the Reference Table Chemistry is used to understand the chemical properties of photochemical systems and to design and optimize photochemical reactions. It helps in predicting the behavior of photochemical systems and in developing new applications.
In thermochemistry, the Reference Table Chemistry is used to understand the chemical properties of thermochemical systems and to design and optimize thermochemical reactions. It helps in predicting the behavior of thermochemical systems and in developing new applications.
In surface chemistry, the Reference Table Chemistry is used to understand the chemical properties of surfaces and to design and optimize surface reactions. It helps in predicting the behavior of surfaces and in developing new applications.
In colloid chemistry, the Reference Table Chemistry is used to understand the chemical properties of colloids and to design and optimize colloidal systems. It helps in predicting the behavior of colloids and in developing new applications.
In supramolecular chemistry, the Reference Table Chemistry is used to understand the chemical properties of supramolecular systems and to design and optimize supramolecular assemblies. It helps in predicting the behavior of supramolecular systems and in developing new applications.
In computational chemistry, the Reference Table Chemistry is used to understand the chemical properties of computational models and to design and optimize computational simulations. It helps in predicting the behavior of computational models and in developing new applications.
In theoretical chemistry, the Reference Table Chemistry is used to understand the chemical properties of theoretical models and to design and optimize theoretical calculations. It helps in predicting the behavior of theoretical models and in developing new applications.
In green chemistry, the Reference Table Chemistry is used to understand the chemical properties of green chemical systems and to design and optimize green chemical reactions. It helps in predicting the behavior of green chemical systems and in developing new applications.
In nanochemistry, the Reference Table Chemistry is used to understand the chemical properties of nanomaterials and to design and optimize nanochemical reactions. It helps in predicting the behavior of nanomaterials and in developing new applications.
In astrochemistry, the Reference Table Chemistry is used to understand the chemical properties of astrochemical systems and to design and optimize astrochemical reactions. It helps in predicting the behavior of astrochemical systems and in developing new applications.
In geochemistry, the Reference Table Chemistry is used to understand the chemical properties of geological systems and to design and optimize geological reactions. It helps in predicting the behavior of geological systems and in developing new applications.
In marine chemistry, the Reference Table Chemistry is used to understand the chemical properties of marine systems and to design and optimize marine chemical reactions. It helps in predicting the behavior of marine systems and in developing new applications.
In atmospheric chemistry, the Reference Table Chemistry is used to understand the chemical properties of atmospheric systems and to design and optimize atmospheric chemical reactions. It helps in predicting the behavior of atmospheric systems and in developing new applications.
In soil chemistry, the Reference Table Chemistry is used to understand the chemical properties of soil systems and to design and optimize soil chemical reactions. It helps in predicting the behavior of soil systems and in developing new applications.
In food chemistry, the Reference Table Chemistry is used to understand the chemical properties of food systems and to design and optimize food chemical reactions. It helps in predicting the behavior of food systems and in developing new applications.
In forensic chemistry, the Reference Table Chemistry is used to understand the chemical properties of forensic systems and to design and optimize forensic chemical reactions. It helps in predicting the behavior of forensic systems and in developing new applications.
In environmental chemistry, the Reference Table Chemistry is used to understand the chemical properties of environmental systems and to design and optimize environmental chemical reactions. It helps in predicting the behavior of environmental systems and in developing new applications.
In materials science, the Reference Table Chemistry is used to understand the chemical properties of materials and to design and optimize material systems. It helps in predicting the behavior of materials and in developing new applications.
In biochemistry, the Reference Table Chemistry is used to understand the chemical properties of biological systems and to design and optimize biological chemical reactions. It helps in predicting the behavior of biological systems and in developing new applications.
In physical chemistry, the Reference Table Chemistry is used to understand the chemical properties of physical systems and to design and optimize physical chemical reactions. It helps in predicting the behavior of physical systems and in developing new applications.
In inorganic chemistry, the Reference Table Chemistry is used to understand the chemical properties of inorganic systems and to design and optimize inorganic chemical reactions. It helps in predicting the behavior of inorganic systems and in developing new applications.
In organic chemistry, the Reference Table Chemistry is used to understand the chemical properties of organic systems and to design and optimize organic chemical reactions. It helps in predicting the behavior of organic systems and in developing new applications.
In polymer chemistry, the Reference Table Chemistry is used to understand the chemical properties of polymer systems and to design and optimize polymer chemical reactions. It helps in predicting the behavior of polymer systems and in developing new applications.
In nuclear chemistry, the Reference Table Chemistry is used to understand the chemical properties of nuclear systems and to design and optimize nuclear chemical reactions. It helps in predicting the behavior of nuclear systems and in developing new applications.
In electrochemistry, the Reference Table Chemistry is used to understand the chemical properties of electrochemical systems and to design and optimize electrochemical chemical reactions. It helps in predicting the behavior of electrochemical systems and in developing new applications.
In photochemistry, the Reference Table Chemistry is used to understand the chemical properties of photochemical systems and to design and optimize photochemical chemical reactions. It helps in predicting the behavior of photochemical systems and in developing new applications.
In thermochemistry, the Reference Table Chemistry is used to understand the chemical properties of thermochemical systems and to design and optimize thermochemical chemical reactions. It helps in predicting the behavior of thermochemical systems and in developing new applications.
In surface chemistry, the Reference Table Chemistry is used to understand the chemical properties of surface systems and to design and optimize surface chemical reactions. It helps in predicting the behavior of surface systems and in developing new applications.
In colloid chemistry, the Reference Table Chemistry is used to understand the chemical properties of colloid systems and to design and optimize colloid chemical reactions. It helps in predicting the behavior of colloid systems and in developing new applications.
In supramolecular chemistry, the Reference Table Chemistry is used to understand the chemical properties of supramolecular systems and to design and optimize supramolecular chemical reactions. It helps in predicting the behavior of supramolecular systems and in developing new applications.
In computational chemistry, the Reference Table Chemistry is used to understand the chemical properties of computational systems and to design and optimize computational chemical reactions. It helps in predicting the behavior of computational systems and in developing new applications.
In theoretical chemistry, the Reference Table Chemistry is used to understand the chemical properties of theoretical systems and to design and optimize theoretical chemical reactions. It helps in predicting the behavior of theoretical systems and in developing new applications.
In green chemistry, the Reference Table Chemistry is used to understand the chemical properties of green chemical systems and to design and optimize green chemical reactions. It helps in predicting the behavior of green chemical systems and in developing new applications.
In nanochemistry, the Reference Table Chemistry is used to understand the chemical properties of nanochemical systems and to design and optimize nanochemical reactions. It helps in predicting the behavior of nanochemical systems and in developing new applications.
In astrochemistry, the Reference Table Chemistry is used to understand the chemical properties of astrochemical systems and to design and optimize astrochemical reactions. It helps in predicting the behavior of astrochemical systems and in developing new applications.
In geochemistry, the Reference Table Chemistry is used to understand the chemical properties of geochemical systems and to design and optimize geochemical reactions. It helps in predicting the behavior of geochemical systems and in developing new applications.
In marine chemistry, the Reference Table Chemistry is used to understand the chemical properties of marine chemical systems and to design and optimize marine chemical reactions. It helps in predicting the behavior of marine chemical systems and in developing new applications.
In atmospheric chemistry, the Reference Table Chemistry is used to understand the chemical properties of atmospheric chemical systems and to design and optimize atmospheric chemical reactions. It helps in predicting the behavior of atmospheric chemical systems and in developing new applications.
In soil chemistry, the Reference Table Chemistry is used to understand the chemical properties of soil chemical systems and to design and optimize soil chemical reactions. It helps in predicting the behavior of soil chemical systems and in developing new applications.
In food chemistry, the Reference Table Chemistry is used to understand the chemical properties of food chemical systems and to design and optimize food chemical reactions. It helps in predicting the behavior of food chemical systems and in developing new applications
Related Terms:
- physical setting chemistry reference table
- reference table chemistry 2011
- nys regents chem reference table
- reference table chemistry 2025
- regents chemistry reference table pdf
- chemistry regents reference table 2025