Understanding the atomic structure of elements is fundamental to grasping the principles of chemistry and physics. One of the most iconic models used to explain the arrangement of electrons in an atom is the Titanium Bohr Model. This model, proposed by Niels Bohr, provides a simplified yet powerful way to visualize the electron configuration of titanium, a transition metal with significant industrial applications. By delving into the Titanium Bohr Model, we can gain insights into the behavior of electrons and the properties of titanium itself.
Understanding the Bohr Model
The Bohr Model, introduced by Niels Bohr in 1913, revolutionized our understanding of atomic structure. It posits that electrons orbit the nucleus in discrete, quantized energy levels, much like planets orbiting the sun. Each energy level, or shell, can hold a specific number of electrons. The model is particularly useful for visualizing the electron configuration of elements with fewer than three electron shells, making it an excellent tool for understanding the Titanium Bohr Model.
The Structure of the Titanium Atom
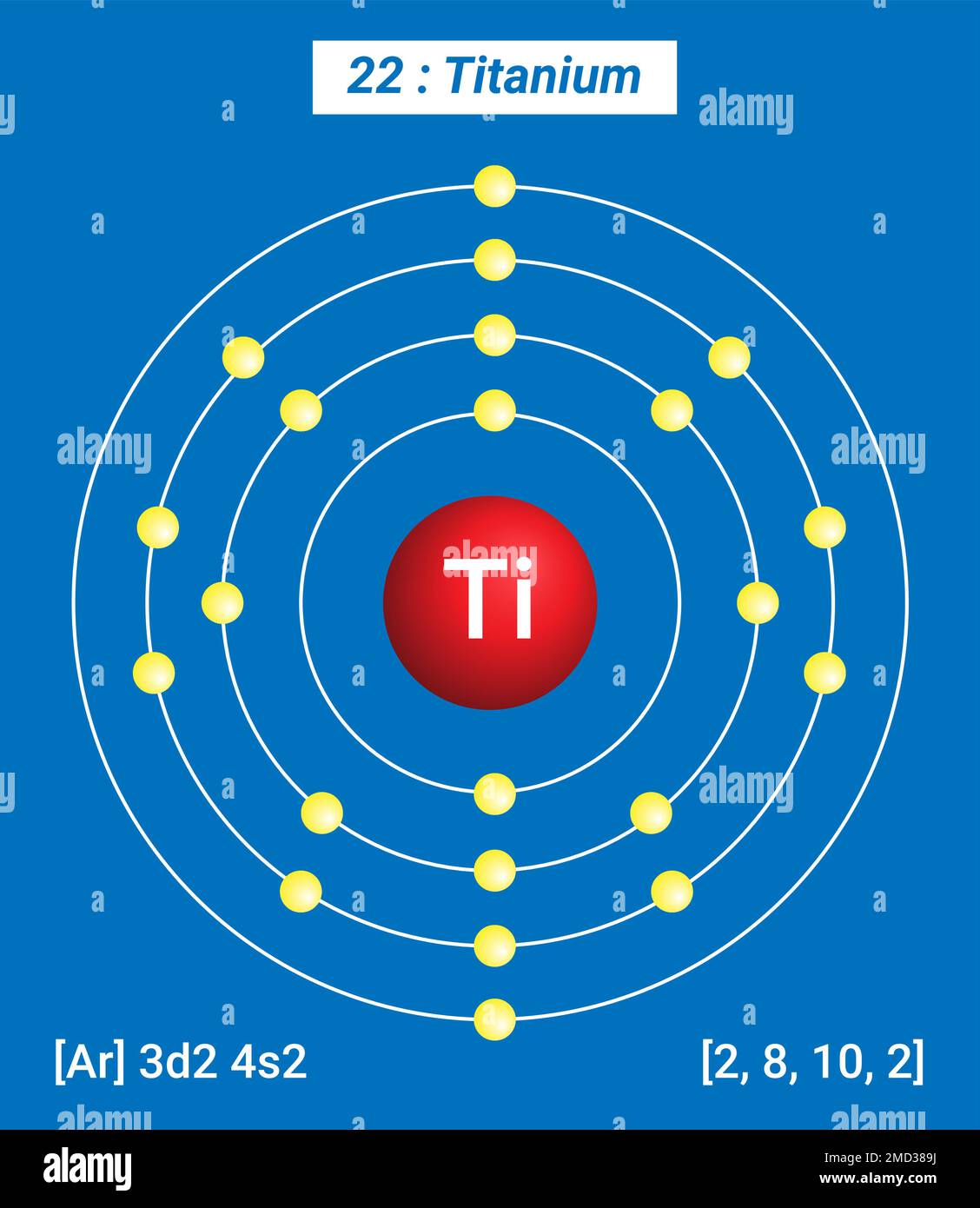
Titanium, with the atomic number 22, has 22 protons and 22 electrons. The electron configuration of titanium can be represented using the Bohr Model, which helps in visualizing the distribution of electrons in different energy levels. The electron configuration of titanium is 2, 8, 10, 2. This means:
- 2 electrons in the first energy level (K shell).
- 8 electrons in the second energy level (L shell).
- 10 electrons in the third energy level (M shell).
- 2 electrons in the fourth energy level (N shell).
This configuration is crucial for understanding the chemical behavior and properties of titanium.
Constructing the Titanium Bohr Model
To construct the Titanium Bohr Model, follow these steps:
- Draw a nucleus at the center of the model. The nucleus contains 22 protons and a number of neutrons equal to the number of protons to maintain neutrality. For titanium, the number of neutrons is approximately 26.
- Draw the first energy level (K shell) around the nucleus. This shell can hold a maximum of 2 electrons. Place 2 electrons in this shell.
- Draw the second energy level (L shell) around the first shell. This shell can hold a maximum of 8 electrons. Place 8 electrons in this shell.
- Draw the third energy level (M shell) around the second shell. This shell can hold a maximum of 18 electrons. Place 10 electrons in this shell.
- Draw the fourth energy level (N shell) around the third shell. This shell can hold a maximum of 32 electrons. Place 2 electrons in this shell.
📝 Note: The Bohr Model is a simplification and does not account for more complex electron behaviors described by quantum mechanics. However, it is a valuable tool for introductory purposes.
Properties of Titanium
Titanium is a transition metal known for its strength, light weight, and resistance to corrosion. These properties make it highly valuable in various industries, including aerospace, automotive, and medical. The Titanium Bohr Model helps in understanding the electron configuration, which in turn influences the chemical and physical properties of the element.
Some key properties of titanium include:
- High strength-to-weight ratio: Titanium is strong yet lightweight, making it ideal for applications where weight is a critical factor.
- Corrosion resistance: Titanium is highly resistant to corrosion, making it suitable for use in harsh environments.
- Biocompatibility: Titanium is biocompatible, meaning it can be used in medical implants without causing adverse reactions.
- High melting point: Titanium has a high melting point, making it useful in high-temperature applications.
Applications of Titanium
Titanium’s unique properties make it indispensable in various fields. Some of the most significant applications include:
- Aerospace: Titanium is used in the construction of aircraft and spacecraft due to its strength and light weight.
- Automotive: Titanium is used in high-performance engines and exhaust systems due to its resistance to heat and corrosion.
- Medical: Titanium is used in medical implants, such as hip replacements and dental implants, due to its biocompatibility.
- Industrial: Titanium is used in chemical processing equipment, marine applications, and sporting goods due to its durability and resistance to corrosion.
Comparing the Bohr Model with Other Atomic Models
The Bohr Model is one of several atomic models used to describe the structure of atoms. While it provides a useful visualization of electron configuration, it has limitations. Other models, such as the Quantum Mechanical Model, offer more accurate descriptions of electron behavior. However, the Titanium Bohr Model remains a valuable tool for introductory purposes and for understanding basic electron configurations.
Here is a comparison of the Bohr Model with other atomic models:
| Model | Description | Strengths | Limitations |
|---|---|---|---|
| Bohr Model | Electrons orbit the nucleus in discrete energy levels. | Simple and easy to visualize. | Does not account for more complex electron behaviors. |
| Quantum Mechanical Model | Electrons exist in probability clouds around the nucleus. | More accurate description of electron behavior. | More complex and difficult to visualize. |
| Shell Model | Electrons are arranged in shells and subshells. | Provides a more detailed description of electron configuration. | Still a simplification compared to quantum mechanics. |
Visualizing the Titanium Bohr Model
Visualizing the Titanium Bohr Model can enhance understanding and retention of the concept. Below is an image that illustrates the electron configuration of titanium according to the Bohr Model.

Educational Significance of the Bohr Model
The Titanium Bohr Model is a valuable educational tool for teaching the basics of atomic structure and electron configuration. It provides a visual representation that helps students understand the arrangement of electrons in different energy levels. This foundational knowledge is essential for more advanced studies in chemistry and physics.
Educators can use the Bohr Model to:
- Introduce the concept of atomic structure and electron configuration.
- Explain the behavior of electrons in different energy levels.
- Demonstrate the relationship between electron configuration and chemical properties.
- Provide a visual aid for understanding more complex atomic models.
In summary, the Titanium Bohr Model offers a simplified yet effective way to visualize the electron configuration of titanium. This model, proposed by Niels Bohr, helps in understanding the basic principles of atomic structure and the properties of titanium. By constructing and visualizing the Titanium Bohr Model, students and educators can gain valuable insights into the behavior of electrons and the unique characteristics of this important transition metal. The Bohr Model, while a simplification, remains a powerful tool for introductory purposes and for laying the groundwork for more advanced studies in chemistry and physics. The applications of titanium, ranging from aerospace to medical implants, highlight the practical significance of understanding its atomic structure. As we continue to explore the properties and applications of titanium, the Titanium Bohr Model will remain a foundational concept in our understanding of atomic behavior.
Related Terms:
- titanium electron configuration chart
- bohr diagram for titanium
- orbital diagram for titanium
- full electron configuration of titanium
- titanium bohr symbol
- full electronic configuration of titanium